Researchers often approach autism spectrum disorder (ASD) through the lens of what might “break down.” While this approach has value, autism is an extremely heterogeneous condition, and diagnosed individuals have a broad range of abilities.
The Gabrieli lab is embracing this diversity and leveraging the strengths of diagnosed individuals by researching their specific “affinities.”
Affinities involve a strong passion for specific topics, ranging from insects to video game characters, and can include impressive feats of knowledge and focus.
The biological basis of these affinities and associated abilities remains unclear, which is intriguing to John Gabrieli and his lab.
“A striking aspect of autism is the great variation from individual to individual,” explains McGovern Investigator John Gabrieli. “Understanding what motivates an individual child may inform how to best help that child reach his or her communicative potential.”

Affinities have traditionally been seen as a distraction “interfering” with conventional teaching and learning. This mindset was upended by the 2014 book Life Animated by Ron Suskind, whose autistic son Owen seemingly lost his ability to speak around age three. Despite this setback, Owen maintained a deep affinity for Disney movies and characters. Rather than extinguishing this passion, the Suskinds embraced it as a path to connection.
Reframing such affinities as a strength not a frustration, and a path to communication rather than a roadblock, caught the attention of Kristy Johnson, a PhD student at the MIT Media Lab, who also has a non-verbal child with autism.
“My interest is in empowering and understanding populations that have traditionally been hard to study, including those with non-verbal and minimally verbal autism,” explains Johnson. “One way to do that is through affinities.”
But even identifying affinities is difficult. An interest in “trains” might mean 18th-century smokestacks to one child, and the purple line of the MBTA commuter rail to another. Serendipitously, she mentioned her interest to Gabrieli one day. He slammed his hands on the table, jumped up, and ran to find lab members Anila D’Mello and Halie Olson, who were gearing up to pursue the neural basis of affinities in autism. A collaboration was born.
Scientific challenge
What followed was six months of intense discussion. How can an affinity be accurately defined? How can individually tailored experiments be adequately controlled? What makes a robust comparison group? How can task-related performance differences between individuals with autism be accounted for?
The handful of studies that had used fMRI neuroimaging to examine affinities in autism had focused on the brain’s reward circuitry. D’Mello and Olson wanted to examine the language network of the brain — a well-defined network of brain regions whose activation can be measured by fMRI. Affinities trigger communication in some individuals with autism (Suskind’s family were using Disney characters to engage and communicate, not simply as a reward). Was the language network being engaged by affinities? Could these results point to a way of tailoring learning for all types of development?
“The language network involves lots of regions across the brain, including temporal, parietal, frontal, and subcortical areas, which play specific roles in different aspects of language processing” explains Olson. “We were interested in a task that used affinities to tap the language network.”
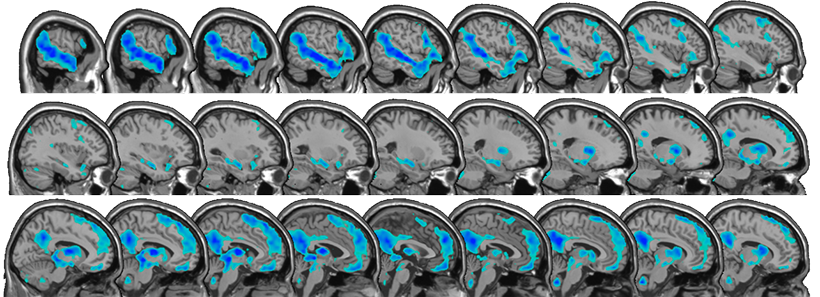
By studying this network, the team is testing whether affinities can elicit “typical” activation in regions of the brain that are sometimes assumed to not be engaged in autism. The approach may help develop better paradigms for studying other tasks with individuals with autism. Regardless of whether there are differences between the group diagnosed with autism and typically developing children, insight will likely be gained into how personalized special interests influence engagement of the language network.
The resulting study is task-free, removing the variable of differing motor or cognitive skill sets. Kids watch videos of their individual affinity in the fMRI scanner, and then listen to stories based on that affinity. They also watch and listen to “neutral” videos and stories about nature that are consistent across all children. Identifying affinities robustly so that the right stimulus can be presented is critical. Rather than an interest in bugs, affinities are often very specific (bugs that eat other bugs). But identifying and cross-checking affinities is something the group is becoming adept at. The results are emerging, but the effects that the team are seeing are significant, and preliminary data suggest that affinities engage networks beyond reward circuits.
“We have a small sample right now, but across the sample, there seems to be a difference in activation in the brain’s language network when listening to affinity stories compared to neutral stories,” explains D’Mello. “The biggest surprise is that the differences are evident in single subjects.”
Future forward
The work is already raising exciting new questions. Are there other brain regions engaged by affinities? How would such information inform education and intervention paradigms? In addition, the team is showing it’s possible to derive information from individualized, naturalistic experimental paradigms, a message for brain imaging and behavioral studies in general. The researchers also hope the results inspire parents, teachers, and psychologists to perceive and engage with an individual’s affinities in new ways.
“This could really help teach us to communicate with and motivate very young and non-verbal kids on the spectrum in a way that is interesting and meaningful to them,” D’Mello explains.
By studying the strengths of individuals with autism, these researchers are showing that, through embracing neurodiversity, we can enhance science, our understanding of the brain, and perhaps even our understanding of ourselves.