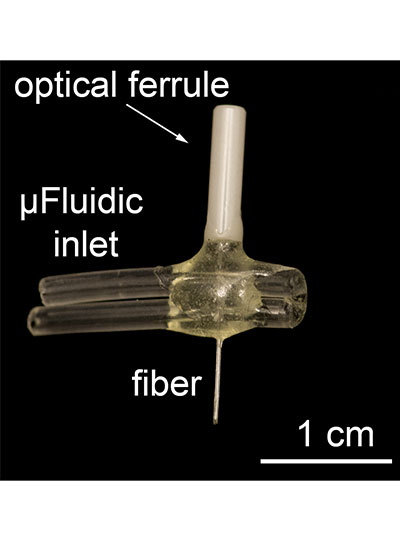
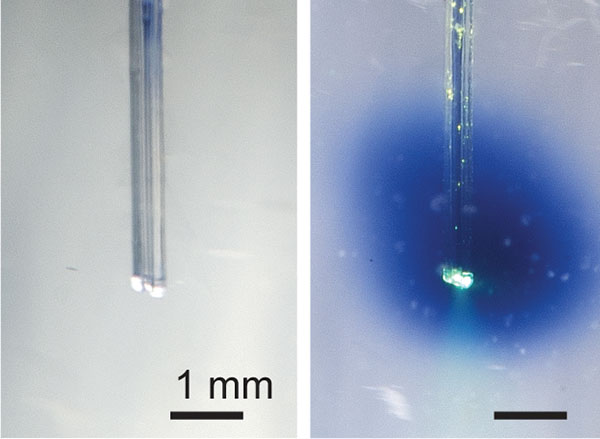
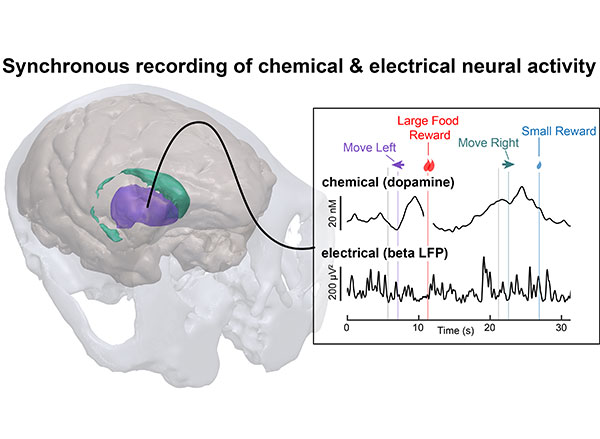
View the interactive version of this story in our Summer 2021 issue of BrainScan.

McGovern Investigator Ed Boyden says his lab’s vision is clear.
“We want to understand how our brains take our sensory inputs, generate emotions and memories and decisions, and ultimately result in motor outputs. We want to be able to see the building blocks of life, and how they go into disarray in brain diseases. We want to be able to control the signals of the brain, so we can repair it,” Boyden says.
To get there, he and his team are exploring the brain’s complexity at every scale, from the function and architecture of its neural networks to the molecules that work together to process information.
And when they don’t have the tools to take them where they want to go, they create them, opening new frontiers for neuroscientists everywhere.
Open to discovery
Boyden’s team is highly interdisciplinary and collaborative. Its specialty, Boyden says, is problem solving. Creativity, adaptability, and deep curiosity are essential, because while many of neuroscience’s challenges are clear, the best way to address them is not. In its search for answers, Boyden’s lab is betting that an important path to discovery begins with finding new ways to explore.
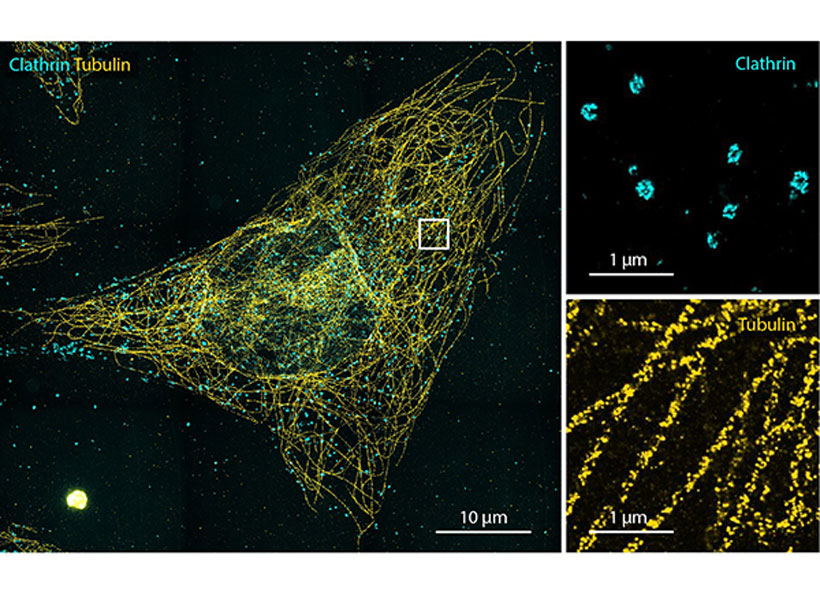
They’ve made that possible with an innovative imaging approach called expansion microscopy (ExM). ExM physically enlarges biological samples so that minute details become visible under a standard laboratory microscope, enabling researchers everywhere to peer into spaces that once went unseen (see video below).
To use the technique, researchers permeate a biological sample with an absorbent gel, then add water, causing the components of the gel to spread apart and the tissue to expand.
This year, postdoctoral researcher Ruixuan Gao and graduate student Chih-Chieh (Jay) Yu made the method more precise, with a new material that anchors a sample’s molecules within a crystal-like lattice, better preserving structure during expansion than the irregular mesh-like composition of the original gel. The advance is an important step toward being able to image expanded samples with single-molecule precision, Gao says.
A revealing look
By opening space within the brain, ExM has let Boyden’s team venture into those spaces in new ways.

In work led by Deblina Sarkar (who is now an assistant professor at MIT’s Media Lab), Jinyoung Kang, and Asmamaw (Oz) Wassie, they showed that they can pull apart proteins in densely packed regions like synapses so that it is easier to introduce fluorescent labels, illuminating proteins that were once too crowded to see. The process, called expansion revealing, has made it possible to visualize in intact brain tissue important structures such as ion channels that help transmit signals and fine-scale amyloid clusters in Alzheimer’s model mice.
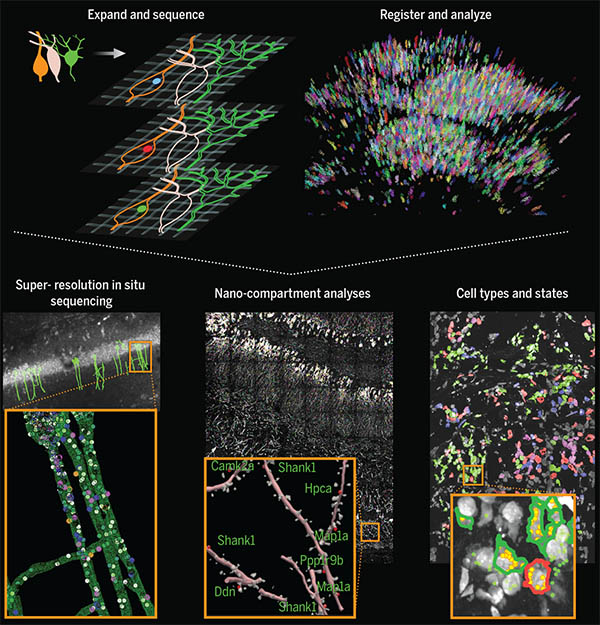
Another reaction the lab has adapted to the expanded-brain context is RNA sequencing—an important tool for understanding cellular diversity. “Typically, the first thing you do in a sequencing project is you grind up the tissue, and you lose the spatial dimension,” explains Daniel Goodwin, a graduate student in Boyden’s lab. But when sequencing reactions are performed inside cells instead, new information is revealed.

Goodwin and fellow Boyden lab members Shahar Alon, Anubhav Sinha, Oz Wassie, and Fei Chen developed expansion sequencing (ExSeq), which copies RNA molecules, nucleotide by nucleotide, directly inside expanded tissue, using fluorescent labels that spell out the molecules’ codes just as they would in a sequencer.
The approach shows researchers which genes are turned on in which cells, as well as where those RNA molecules are—revealing, for example, which genes are active in the neuronal projections that carry out the brain’s communications. A next step, Sinha says, is to integrate expansion sequencing with other technologies to obtain even deeper insights.
That might include combining information revealed with ExSeq with a topographical map of the same cells’ genomes, using a method Boyden’s lab and collaborators Chen (who is now a core member of the Broad Institute) and Jason Buenrostro at Harvard have developed for DNA sequencing. That information is important because the shape of the genome varies across cells and circumstances, and that has consequences for how the genetic code is used.
Using similar techniques to those that make ExSeq possible, graduate students Andrew Payne, Zachary Chiang, and Paul Reginato figured out how to recreate the steps of commercial DNA sequencing within the genome’s natural environment.
By pinpointing the location of specific DNA sequences inside cells, the new method, called in situ genome sequencing (IGS) allows researchers to watch a genome reorganize itself in a developing embryo.
They haven’t yet performed this analysis inside expanded tissue, but Payne says integrating in situ genome sequencing (IGS) with ExM should open up new opportunities to study genomes’ structure.
Signaling clusters
Alongside these efforts, Boyden’s team is working to give researchers better tools to explore how molecules move, change, and interact, including a modular system that lets users assemble sets of sensors into clusters to simultaneously monitor multiple cellular activities.
Molecular sensors use fluorescence to report on certain changes inside cells, such as the calcium that surges into a neuron after it fires. But they come in a limited palette, so in most experiments only one or two things can be seen at once.
Graduate student Shannon Johnson and postdoctoral fellow Changyang Linghu solved this problem by putting different sensors at different points throughout a cell so they can report on different signals. Their technique, called spatial multiplexing, links sensors to molecular scaffolds designed to cling to their own kind. Sensors built on the same scaffold form islands inside cells, so when they light up their signals are distinct from those produced by other sensor islands.
Eventually, as new sensors and scaffolds become available, Johnson says the technique might be used to simultaneously follow dozens of molecular signals in living cells. The more precise information they can help people uncover, the better, Boyden says.
“The brain is so full of surprises, we don’t know where the next big discovery will come from,” he says. With new support from the recently established K. Lisa Yang and Hock E. Tan Center for Molecular Therapeutics in Neuroscience, the Boyden lab is positioned to make these big discoveries.
“My dream would be to image the signaling dynamics of the brain, and then perturb the dynamics, and then use expansion methods to make a map of the brain. If we can get those three data sets—the dynamics, the causality, and the molecular organization—I think stitching those together could potentially yield deep insights into how the brain works, and how we can repair it in disease states.”



 View the interactive version of this story in our
View the interactive version of this story in our  Taking advantage of its pernicious spread, neuroscientists use a modified version of the rabies virus to introduce a fluorescent protein to infected cells and visualize their connections (above). As a graduate student in Edward Callaway’s lab at the Salk Institute for Biological Studies, Wickersham figured out how to limit the virus’s passage through the nervous system, allowing it to access cells that are directly connected to the neuron it initially infects, but go no further. Rabies virus travels across synapses in the opposite direction of neuronal signals, so researchers can deliver it to a single cell or set of cells, then see exactly where those cells’ inputs are coming from.
Taking advantage of its pernicious spread, neuroscientists use a modified version of the rabies virus to introduce a fluorescent protein to infected cells and visualize their connections (above). As a graduate student in Edward Callaway’s lab at the Salk Institute for Biological Studies, Wickersham figured out how to limit the virus’s passage through the nervous system, allowing it to access cells that are directly connected to the neuron it initially infects, but go no further. Rabies virus travels across synapses in the opposite direction of neuronal signals, so researchers can deliver it to a single cell or set of cells, then see exactly where those cells’ inputs are coming from. In Feng’s lab, research scientist Martin Wienisch is working to make it easier to control this aspect of delivery. Rather than relying on the genetic makeup of an entire animal to determine where a virally-transported gene is switched on, instructions can be programmed directly into the virus, borrowing regulatory sequences that cells already know how to interpret.
In Feng’s lab, research scientist Martin Wienisch is working to make it easier to control this aspect of delivery. Rather than relying on the genetic makeup of an entire animal to determine where a virally-transported gene is switched on, instructions can be programmed directly into the virus, borrowing regulatory sequences that cells already know how to interpret. Enhancers will also be useful for delivering potential gene therapies to patients, Wienisch says. For many years, the Feng lab has been studying how a missing copy of a gene called Shank3 impairs neurons’ ability to communicate, leading to autism and intellectual disability. Now, they are investigating whether they can overcome these deficits by delivering a functional copy of Shank3 to the brain cells that need it. Widespread activation of the therapeutic gene might do more harm than good, but incorporating the right enhancer could ensure it is delivered to the appropriate cells at the right dose, Wienisch says.
Enhancers will also be useful for delivering potential gene therapies to patients, Wienisch says. For many years, the Feng lab has been studying how a missing copy of a gene called Shank3 impairs neurons’ ability to communicate, leading to autism and intellectual disability. Now, they are investigating whether they can overcome these deficits by delivering a functional copy of Shank3 to the brain cells that need it. Widespread activation of the therapeutic gene might do more harm than good, but incorporating the right enhancer could ensure it is delivered to the appropriate cells at the right dose, Wienisch says.